Department of Diagnostic Imaging, Graduate School of Medicine, Hokkaido University
Kohsuke Kudo, M.D., Ph.D. Internal
Medicine
- Education:
-
- 1989–1995
- Hokkaido University School of Medicine, Awarded the MD degree
- 1999-2003
- Hokkaido University Graduate School of Medicine, Awarded the PhD degree
- Professional experience:
-
- 1995-1997
- Resident, Radiology, Hokkaido University Hospital
- 1997-1999
- Director, Central CI Clinic
- 2003-2004
- Fellow, Radiology, Hokkaido University Hospital
- 2004-2006
- Instructor, Radiology, Hokkaido University Graduate School of Medicine
- 2006-2007
- Visiting Assistant Professor, MR Research, Wayne State University
- 2007-2008
- Instructor, Radiology, Hokkaido University Hospital
- 2008-2013
- Assistant Professor, Iwate Medical University
- 2013
- Associate Professor, Hokkaido University Hospital
- 2016
- Clinical Professor, Hokkaido University Hospital
- 2019
- Current Position
- Current position:
- Professor
Department of Diagnostic Imaging, Hokkaido University Graduate School of Medicine
To develop a novel image analysis technique and an MRI contrast agent using in-house computer programs

The advancement and sophistication of medicine have accelerated the development of diagnostic imaging technology, such as computed tomography (CT), magnetic resonance imaging (MRI) and positron emission tomography (PET). Against this background, Professor Kohsuke Kudo, who directs the Department of Diagnostic Imaging at the Hokkaido University Graduate School of Medicine and two clinical departments[1] at Hokkaido University Hospital, has promoted the R&D and commercialization of diagnostic imaging technology to scan the brain, spinal cord and other areas of the body using software he has designed and programmed, and he has played a part in driving innovations in multimodal diagnostic imaging technology, which integrates multiple imaging technologies.
While in graduate school, Professor Kudo decided to take advantage of computer programming, a pastime during his days at medical school, in his research, and he authored software to facilitate the high-speed image analysis of cerebral blood flow (perfusion) by combining CT and MRI techniques. The software was released as the freeware Perfusion Mismatch Analyzer (PMA) in 2006, and its successor, PMAneo, obtained pharmaceutical approval in 2019. This software has served as a platform for his image-processing research.
In addition, he embarked on a study to visualize cerebral oxygen metabolism using magnetic susceptibility (a measure of the extent to which a material magnetizes in response to an applied magnetic field), which is a fundamental theory in MRI. He devised an algorithm that converts the amount of cerebral deoxygenated hemoglobin, which varies depending on conditions such as blood flow, hyperventilation and pharmacological stress, into magnetic susceptibility and maps changes in the oxygen extraction fraction (oxygen metabolism).
With an eye to applying this analytical technique, which is based on magnetic susceptibility, to Alzheimer dementia diagnosis, in 2016 Professor Kudo undertook research in conjunction with Hitachi, Ltd. to develop new MRI imaging and analytical methods that would significantly reduce scan time and thus enable the early diagnosis of the disease.
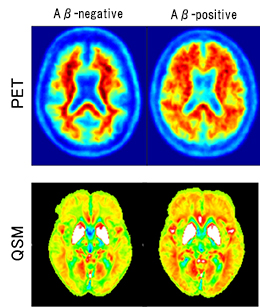
They developed a new analytical technique called quantitative susceptibility mapping (QSM), which quantifies the magnetic susceptibility of the iron that binds to amyloid beta, an abnormal protein that is deposited in the brains of those with Alzheimer dementia, and visualizes the state of iron deposition. By combining QSM with voxel-based morphometry (VBM), a traditional diagnostic technique for the objective assessment of brain atrophy, they developed a fast and patient-friendly MRI diagnostic technique. “With the traditional VBM technique alone, it’s difficult to diagnose dementia before its onset,” Professor Kudo says. “But the finding of iron deposition, which precedes the onset of symptoms, leads to the early diagnosis of the disease.” (Fig.1)
Using safe and secure "Water-17O"/17O-labeled water (an MRI contrast agent) to visualize water dynamics in the body
Professor Kudo has dedicated himself to developing a new diagnostic imaging technology that uses oxygen-17 (17O), a stable isotope that is harmless to humans and is the only oxygen isotope capable of generating an MRI signal. He is focusing on using 17O as an MRI contrast agent (17O-labeled water, a tracer of water molecules) to visualize water distribution in body tissues.
Specializing in the diagnostic imaging of the brain and spinal cord, Professor Kudo has undertaken a study in which 17O-labeled water (water-17O) is administered to dementia patients and others for the multiscale observation of water dynamics (the dynamics of water molecules) in the glymphatic system, which like lymphatic system, clears the wastes and debris that build up between cells. The multiscale observation includes an isotope microscope system and a macroscopic MRI system.
“Thanks to the blessed environment at Hokkaido University, we were able to develop the nation’s first MRI contrast agent and publish research findings on water-17O that are highly original for anywhere in the world,” Professor Kudo says. “With the cooperation of the Hokkaido University Hospital Clinical Research and Medical Innovation Center , which opened in 2014 to develop cutting-edge medical technologies, we developed a water-17O product called PSO17, which is a GMP sterile product that can be safely administered to humans. A great advantage is the way it enables the direct observation of how water moves in and out of cells at the microscopic level using an isotope microscope system[2] that’s available only at CRIS—Hokkaido University’s Creative Research Institution Sousei. CRIS also solved a problem unique to microscopy—the evaporation of water from samples in a vacuum environment—by developing a frozen section technique.”
Toward gaining approval for the pharmaceutical use of water-17O, Professor Kudo became the first to enlist the help of the Phase I Clinical Trial Support Division (a.k.a. the Phase Ⅰ Unit) at the Clinical Research and Medical Innovation Center. He conducted an investigator-initiated phase I trial in healthy adults and has already begun a phase II clinical trial. “We’ve gradually begun to administer water-17O to patients because it’s become clear that it’s safe for healthy people. I hope we can establish its usefulness for patients with dementia, hydrocephalus and other diseases, toward obtaining pharmaceutical approval within several years. And given its potential application to other organs and diseases and to innovative therapeutic strategies, we’re also working to form partnerships with other departments and universities as part of our efforts to integrate different fields of study.”
Ms. Yumi Nakada, a first-year master’s student, is engaged in research on water-17O together with Assistant Professor Hiroyuki Kameda. Dr. Kameda says, “I’ve felt that the currently available analytical methods are inadequate to answer some of the clinical questions we have when we interpret images of the central nervous system. Since it’s difficult to trace water dynamics, it’s truly fascinating to be able to use an MRI contrast agent that allows such tracing without any harm to humans.” Ms. Nakada majored in radiological technology in the Hokkaido University Faculty of Health Sciences and has conducted research on functional MRI of the brain. She expressed her aspiration to continue her studies while obtaining the latest information about MRI under Professor Kudo.
Professor Kudo calls on students with an interest in diagnostic imaging or AI to join him in R&D, saying: “R&D continues to make rapid progress, as AI technologies are embedded in every process of diagnostic imaging, including pre-test scheduling, image resolution enhancement, the automatic detection of pathological changes in images, and diagnoses. Our lab covers the organs in the whole body and covers all our imaging modalities, based on our multi-organ, multi-modal perspective. The lab members are engaged in research in myriad fields based on their interests. I hope students will first and foremost acquire a thorough knowledge of diagnostic imaging of the whole body and then find areas of interest toward which to direct their academic pursuits.”
Professor Kudo continues: “A project to develop human resources for AI research that was launched by Tohoku University, which is the main institution, with participation by Hokkaido University and Okayama University was selected in September 2020 for funding by the Ministry of Education, Culture, Sports, Science and Technology (MEXT), so we’re planning to consolidate AI research in the healthcare field to develop human resources who can help advance AI for medical use. I can hardly wait to hear all the great ideas for using AI technology to address medical needs.”

[1] ^ The Department of Diagnostic and Interventional Radiology , which uses CT (computed tomography) and MRI (magnetic resonance imaging) to diagnose illnesses and which provides treatments using IVR (interventional radiology); and the Department of Nuclear Medicine , which uses PET (positron emission tomography) and SPECT (single photon emission computed tomography) to perform nuclear medicine diagnosis and therapy
[2] ^ Developed by Professor Hisayoshi Yurimoto of the Faculty of Science, Hokkaido University, and other researchers, this is the world’s only isotope microscope system to analyze particles from asteroid 25143 Itokawa which were brought to Earth in 2010 by the asteroid probe Hayabusa. It is the first such system to enable the imaging (visualization) of stable isotopes. The system is also being used to analyze the sand from asteroid 162173 Ryugu, which was brought to Earth in December 2020 by Hayabusa 2.
(Interviewed in October 2020)
Offering visiting lectures at high schools to convey the allure of diagnostic imaging and AI for medical use

In the Academic Fantasista, an annual event in which Hokkaido University researchers in various fields offer visiting lectures at high schools, Professor Kudo and Associate Professor Kenji Hirata lecture about topics including MRI, the mechanism of cerebral diagnostic imaging, AI for medical use, and the research projects they are engaged in. “Since some students have already used AI in a free development environment or have received an MRI test themselves, we always have lively Q&A sessions,” Professor Kudo says with a glow. “In fact, I give lectures to students with the hidden agenda of ‘harvesting rice while it’s still green,’ trying to nudge any students who are curious about diagnostic imaging toward my lab.”